
15 September, 2020


Unique star-shaped polymers have highly tunable properties that will open up a range of new applications.
Plastics are ubiquitous in modern life, used in packaging, computers, medical equipment and many other devices. All of these applications require different material properties, which depend on the nature of the polymer molecules within the plastics.
KAUST researchers are at the forefront of efforts to develop unusual star-shaped polymers that could open up fresh opportunities for these materials. “Because the heart of a material’s properties is at the molecular level, this unique structure gives the materials unique properties and, therefore, applications,” says Distinguished KAUST Professor Nikos Hadjichristidis.
Polymers are built from repeating chemical units called monomers. The identity of the monomer influences a material’s physical properties, but so too does the arrangement of the polymer chains and the interactions among them. By changing a polymer’s architecture, researchers can fine tune important characteristics, such as elasticity and density.
In 1992, Hadjichristidis and his Ph.D. student Hermis Iatrou, now at the University of Athens, Greece, created an entirely new polymer architecture with several different types of polymers all meeting at a point1. Hadjichristidis called them miktoarm star polymers (from the Greek word µικτός, meaning "mixed"). Because they combine different polymers, they are highly tunable, and the discovery spawned a research field that has yielded hundreds of papers.
In their latest research, Hadjichristidis and his colleagues developed a better way to incorporate polyethylene (PE) into miktoarm star polymers2. PE-based materials have a wide range of applications thanks to their excellent properties: they are inexpensive, stable to heat, strong and flexible. Consequently, miktoarm polymers that contain PE might offer a way to combine the tunability of star polymers with the versatility of PE.
The method previously used to make PE-based star polymers led to stars having PE arms with very short branches, which made the material less robust. So Hadjichristidis’ team developed an improved approach that combines two different polymerization techniques.
They built the stars around a molecule called 4-(dichloromethylsilyl)-diphenylethylene, which contains chemical groups that act as anchor points for polymer chains. The researchers hooked two polyisoprene (PI) chains onto this foundation and then grew a third arm from a different anchor point. By choosing different monomers, the star’s third arm could either be PI, polystyrene or polybutadiene.
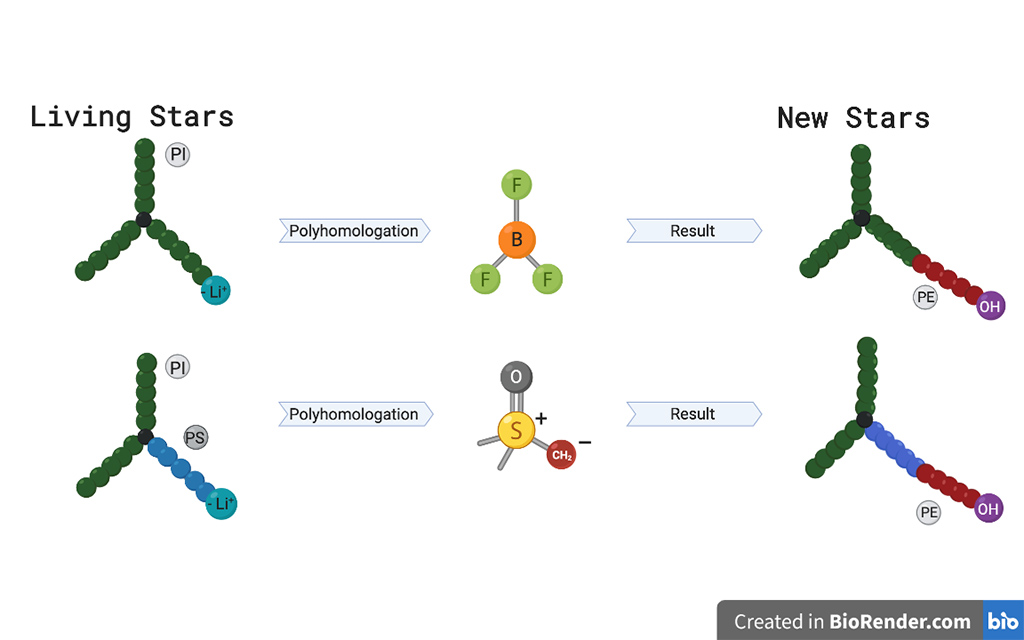
The researchers then added a boron reagent that bonded to the tip of this third arm. Another reagent inserted repeating monomer units between the boron atom and the star, creating a section of PE. Once each star was released from the boron atom, this perfectly linear PE chain contained no short branches and was capped by a hydroxyl group.
"By comparing three types of PE-based stars, the researchers could study how the different structures affected their physical properties, such as self-assembly and crystallinity," says Konstantinos Ntetsikas, who led the experimental work at KAUST. “The next step is to synthesize more PE-based star polymers with varied compositions and study their self-assembly behavior,” he says.
The researchers plan to incorporate polymers that make the materials more soluble, for example, to compare how the stars behave in solution. The hydroxyl group at the end of the PE arm could also be used to attach more polymer molecules onto the star, producing even more complex architectures with unprecedented properties.
“With the knowledge achieved from this and other studies, we can design and synthesize the polymers to precisely achieve targeted material properties,” says Edwin L. Thomas at Rice University in Houston, Texas, who collaborated with Hadjichristidis’s team on the research. “Our future work will be aimed at finding processing conditions that will enable the PE arms to have much higher crystallinities, even above 90 percent.”
Hadjichristidis and his KAUST colleague Yves Gnanou hope to incorporate biodegradable sections into miktoarm stars that could make them useful in biomedical applications or add fluorinated polymers so that they might be applied in manufacturing electronic devices. “Their unique microstructures will solve problems otherwise impossible with their linear counterparts,” Hadjichristidis says.
The researchers also aim to develop processes that are more amenable to manufacturing miktoarm star polymers. For now, the methods used to build the new stars are very sensitive to air and moisture, so they must be carried out in high-vacuum conditions. “So far, the easy and cost-effective synthesis of well-defined miktoarm stars remains a challenge,” says Hadjichristidis. “These challenges will undoubtedly inspire more research in the future.”