Aromaticity, a concept usually used to explain the striking stability and unusual reactivity of certain carbon-based molecules, could inspire the design of new catalysts with novel uses, KAUST researchers have shown.
Chemists first came upon the anomalous behavior of aromatic molecules in the nineteenth century while studying benzene. The unexpected stability of this six-carbon cyclic structure comes down to its electrons.
In general, bonding electrons hold a specific pair of atoms together in a discrete chemical bond. But in benzene, six electrons form a delocalized ring across the molecule. A host of other molecules share this feature. “Many classic examples of organic and organometallic reactivity can be explained on this basis,” says Théo Gonçalves, a research scientist in Kuo-Wei Huang’s lab. “But although the concept is well known, there are limited practical chemical applications of aromaticity,” he adds.
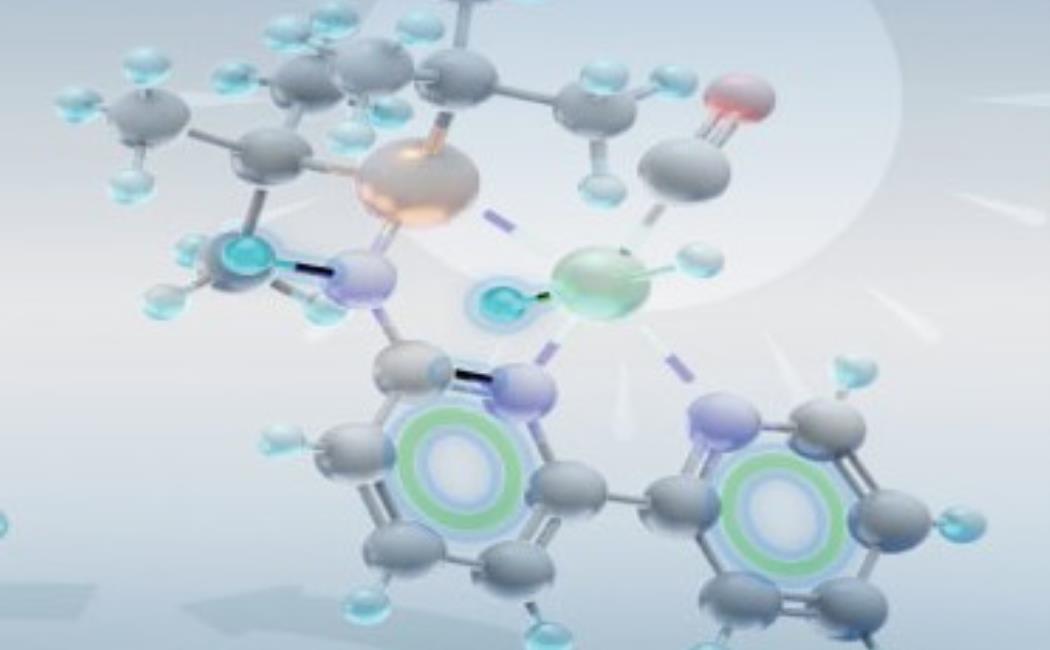
One area of practical application is in the field of catalysis. The Huang group recently developed an unusual family of catalysts called PN3(P) pincer complexes. In most catalysts, the central metal ion is where all the bond breaking and making takes place. In PN3(P) complexes, the pincer ligands around the metal can also be active participants in the catalytic process. “Our PN3(P) ligand platform enables catalytic applications beyond conventional systems where metal is the center of reactivity,” Huang says.
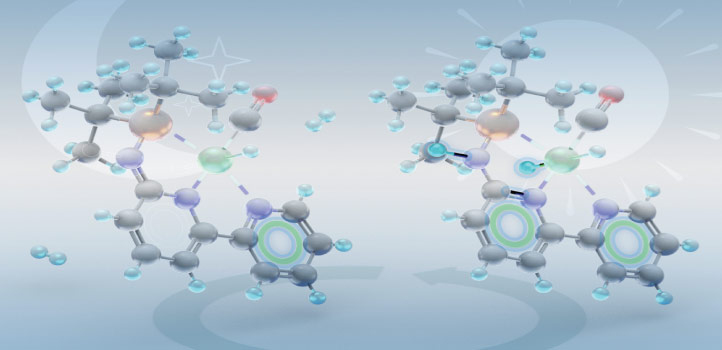
As the team studied pincer complexes’ catalytic behavior, they showed that a six-membered ring structure transiently forms during catalysis and that aromaticity was coming into play. “We have provided strong evidence that during the catalytic cycle, our catalysis benefits from the additional energy coming from aromatization of the ring,” Gonçalves says. “Tuning the degree of aromatization will gently tune the reaction output.”
The PN3(P) pincer family possesses high catalytic performance for reactions such as the selective hydrogen production from formic acid for reducing carbon dioxide (CO2) and for forming esters and imines. But the real value of the research could be from the new insights it generates into the role of aromaticity in catalysis, and the new horizons that open up as a result. “Prior to our work, the importance of the aromaticity was not highlighted in this field,” Gonçalves says. “Fundamentally understanding aromatization and dearomatization will allow redesign of catalysts towards better performance and perhaps novel reactivity.”
“Our discovery is not about identifying a new or better catalyst for a known reaction, but about opening a new field for unlimited new opportunities in the future,” Huang adds.